Lincotek has received FDA 510(k) clearance for its SportLinc Syndesmosis device, a sterile single-use implant designed to stabilize syndesmotic ankle injuries, known as high ankle sprains. This milestone allows the company to target the orthopedic original equipment manufacturer market, offering a specific solution for a common injury in athletes.
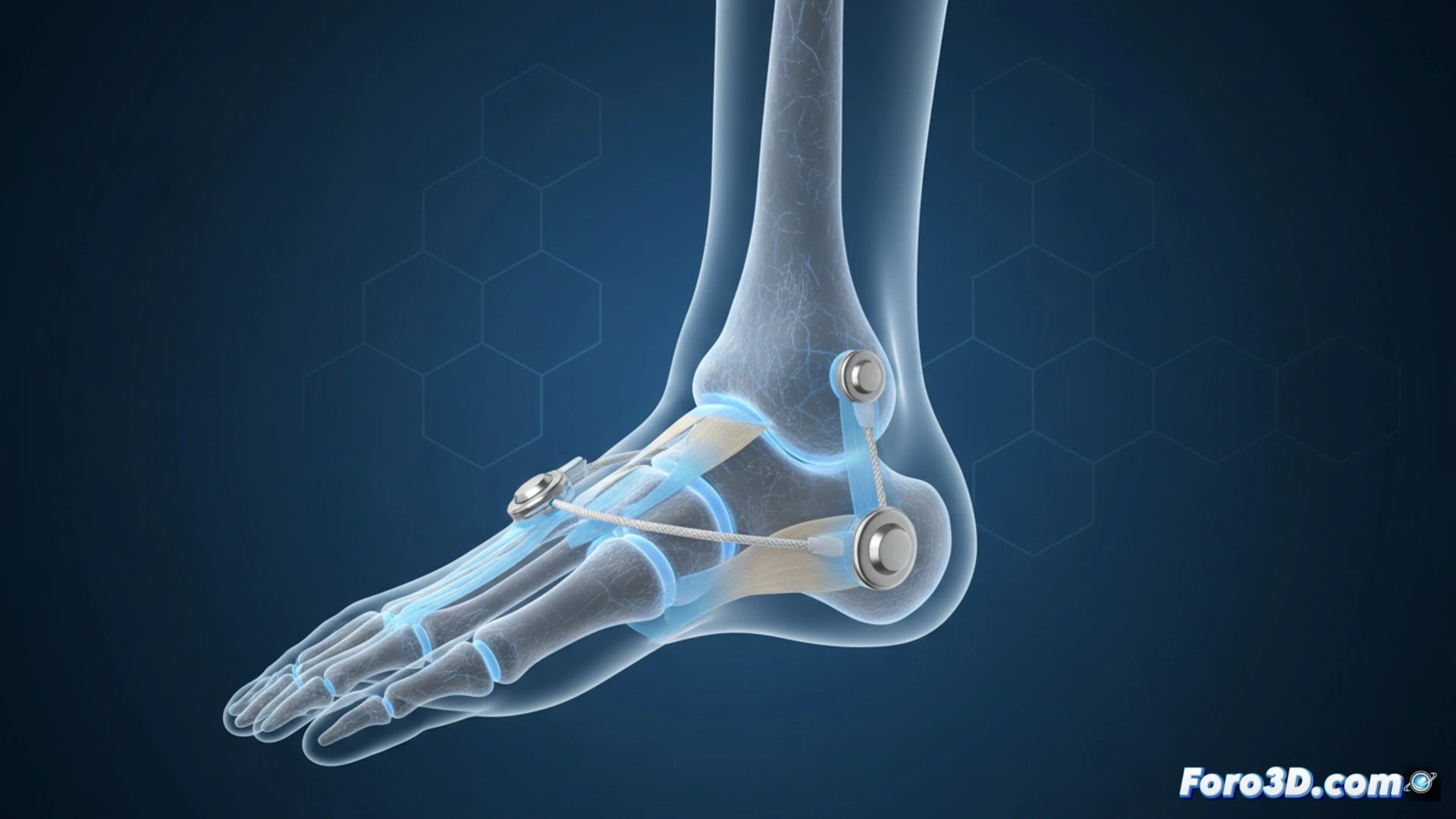
Design and Function of the SportLinc Implant 🦴
The SportLinc Syndesmosis is a sterile single-use implant designed to fix the distal tibiofibular joint following a syndesmotic injury. Its design aims to simplify the surgical procedure by eliminating the need to prepare the implant in the operating room. As a ready-to-use device, it reduces steps during surgery, which can influence operative times and standardize the fixation process.
The Ankle That Didn't Want Common Sprains 🏃
Finally, high ankle sprains have a worthy adversary: an implant that comes in its own sterile packaging, like a bone fixation snack. Because, let's be honest, when a ligament decides to be difficult and tear in the upper part, it's time to bring out the heavy artillery. Bandages and rest are no longer enough; now you need a device with a sports team name to remind your ankle who's in charge.